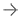William van Kampen, Xoran Chief Technology Officer and principal investigator on the project.
June 24, 2022 —Xoran Technologiesannounced they have begun work onPhase 2of their mobile lung grant with the goal to confirm the safety and utility of a future thoracic point-of-careCT system支持FDA 510k提交。
“It’s hard to overstate how transformative this technology would be for us in the ICU,” saysRobert Dickson, MD, Associate Professor—Pulmonary & Critical Care Medicine, Associate Professor—Microbiology & Immunology, and a clinical collaborator, all at the University of Michigan. “Every day, we make clinical decisions based on chest X-rays, which are limited in what they can tell us about what is going on in the chest or abdomen. Our patients are often too sick to transport down to radiology, or they have a communicable disease likeCOVID-19that we don’t want to spread around the hospital. A bedside scanner would have immediate impact in how we manage our sickest patients.”
In 2021, Xoran Technologies was notified of a grant award from theNational Heart, Lung, and Blood Institute(NHLBI) through theNational Institutes of Health(NIH) to support the company’s research and development efforts for lung cone beamcomputed tomography(CT) imaging. Earlier this year, Xoran announced they completed Phase 1 of this grant. Just last month, Xoran submitted its 510k forTRON, an open-bore fluoroscopy CT for full-body point-of-care imaging.
“We greatly appreciate the support from NHLBI as well as a fantastic collaboration with doctors at the University of Michigan,” statesWilliam van Kampen他是Xoran公司的首席技术官,也是该项目的首席研究员。“这项资助使我们能够获得宝贵的临床反馈,加速这项令人兴奋的技术的商业化,并帮助将成像技术带给有极大需求的患者。”
This project allows Xoran to optimize a point-of-care solution specifically forlung imaging. The combined Xoran and UofM teams aim to develop a highly deployable CT scanner intended for use in ICU, especially for patients withacute respiratory failurerequiring mechanical ventilation.
For more information:www.xorantech.com


 August 08, 2022
August 08, 2022