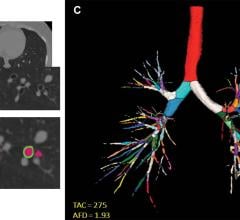
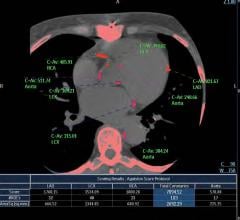
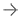3D系统公司的D2P软件获得fda批准,允许临床医生3D - d打印诊断患者特异性解剖模型。图片由3D系统提供。
September 12, 2019 — 3D Systems has received additional U.S. Food and Drug Administration (FDA) 510(k) clearance for its D2P software allowing clinicians to3-D printdiagnostic patient-specific anatomic models.
D2P (DICOM-to-Print) relies on automatic segmentation tools driven by deep learning that allow medical practitioners to quickly create accurate,digital 3-D anatomic modelsfrom medical imaging data. With the additional FDA clearance, D2P addresses the growing demand by point-of-care (POC) institutions for in-house manufacturing using an accurate and reliable 3-D segmentation solution that can produce 3-D-printed models.
D2P现在还包括深度学习图像处理技术和虚拟现实(VR)可视化的最新进展。这使得医院和设备制造商大大减少了与创建3d模型相关的时间。
该软件还包括一个体积虚拟现实解决方案,能够在三维环境中即时查看患者的扫描-促进手术计划和医务人员与患者之间的对话。
"We are used to going into surgery with uncertainties and an arsenal of contingency plans," saidSolomon Dadia, M.D., deputy director of the orthopedic-oncology department and director of the 3D surgical center at Souraski Medical Center in Tel-Aviv. "With 3-D-printed models and enhanced 3-D visualization tools such as VR, we are able to gain a better understanding of the surgery and pathology we are going to treat. This allows us to come up with a more precise surgery plan designed to deliver a better surgical outcome."
In accordance with the FDA announcement on new guidelines for 3-D-printed, patient-specific anatomic models in 2017, diagnostic-quality models must be an output of a Class II regulated medical device software. According to the company, 3D Systems is the only company to offer both a software solution and compatible printers of its own that meet this regulatory requirement. Anatomic models can be produced using a variety of 3D Systems printing technologies — ColorJet Printing, MultiJet Printing, Stereolithography and Selective Laser Sintering — including materials that are capable of sterility and biocompatibility.
For more information:www.3dsystems.com


 August 09, 2022
August 09, 2022