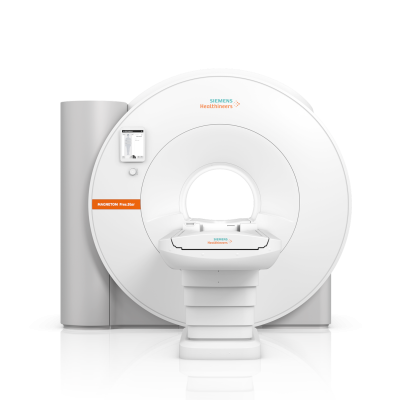
July 5, 2022 —Siemens Healthineershas announced the Food and Drug Administration (FDA) clearance of theMagnetom Free.Star, a cost-effective whole-body magnetic resonance (MR) scanner designed to help improve patient access toMRI. The second scanner on the High-V MR platform following theMAGNETOM Free.Max, the Magnetom Free.Star has a 60 cm patient bore and combines a 0.55 Tesla (0.55T) field strength with deep learning technologies and advanced image processing.
Magnetom免费的。Star是该公司最便宜的核磁共振扫描仪,3.3吨,不到80英寸高,是其有史以来最小的,最轻的全身核磁共振系统。它只需要不到1升的液氦,而且没有淬火管,有助于减少基础设施和生命周期成本。The scanner’s reducedenergy consumptioncontributes to a reduction in total lifecycle costs of more than 30 percent compared to conventional scanners.Deep Resolvealgorithms perform targeted denoising and employ deep learning to deliver sharp, high-resolution images, elevating image quality to a level previously achievable only using MR scanners with much higher field strengths. ThemyExam Companionworkflow solution leverages artificial intelligence to help the user conduct a more efficient patient examination.
“Siemens Healthineers believes that patients everywhere deserve access to magnetic resonance imaging and its unique benefits,” saidJane Kilkenny他是西门子健康工程师北美公司磁共振业务副总裁。“Magnetom自由。Star进一步证明了我们的坚定承诺,即为客户提供更经济、更容易操作、更容易安装在美国各种医疗机构的核磁共振扫描仪。”
For more information:www.siemens-healthineers.com


 August 11, 2022
August 11, 2022