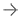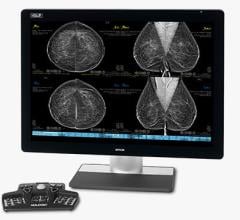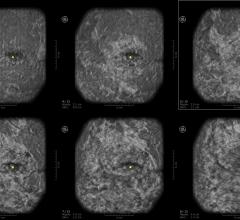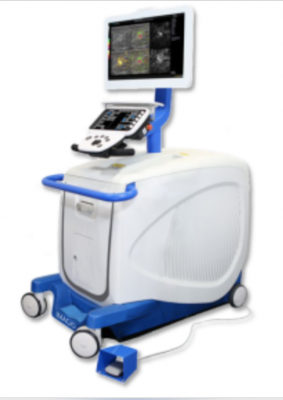
The Imagio Breast Imaging System combines laser light and sound with conventional ultrasound technology to provide fused functional and anatomic images (referred to as optoacoustic/ultrasound or OA/US) in real time, increasing confidence in diagnostic accuracy.
April 23, 2021 — The U.S. Food and Drug Administration (FDA) has approved Seno Medical'sImagio P200003 Breast Imaging System.
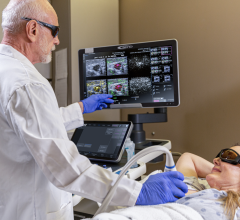
The Imagio Breast Imaging System uses bothoptoacoustic(OA) andultrasound(US) to image breast tissues to help physicians examine breastlesions.Imagio乳腺成像系2022世界杯篮球预选赛赛程统具有软件和硬件,可生成OA图像和常规超声图像的乳房。首先,超声模式最初用于评估任何临床或影像学关注的病灶区域。对于超声BI-RADS 3-5肿块,与单独超声相比,使用OA图像可以改善分类。The OA mode is not indicated for ultrasoundBI-RADS1 and 2 findings. The Imagio® Breast Imaging System includes anartificial intelligence(AI) based software function to assist the users with BI-RADS classifications.
The Imagio Breast Imaging System is used when a physician finds an unusual area of breast tissue usingmammographyandultrasound.使用该设备生成的OA图像用于评估乳腺病变。与单独的超声图像相比,Imagio乳腺成像系统生成的OA图像可以提供更多关2022世界杯篮球预选赛赛程于乳腺成像区域的信息,如结构和功能(如血氧水平)。超声BI-RADS 3-5肿块的附加信息可以帮助医生评估乳腺病变和BI-RADS分类。
The Imagio Breast Imaging System should not be used in people who:
- Are pregnant
- Have open sores like insect bites, rash, poison ivy, or scratches on the skin on the side of the breast)
- Are experiencing effects like phototoxicity (irritation due to light) because of taking drugs such assulfonamides,ampicillin,tetracyclinewithin the last 72 hours
- Are going throughphototherapy
- Have a history of any photosensitive disease (e.g.,porphyria,lupus erythematosus)
- Are being treated for a photosensitive disease or are experiencing photosensitivity
For more information:https://senomedical.com


 August 11, 2022
August 11, 2022